Role of Dendrimers in drug delivery for Cancer therapy
Abstract
In the history of nanotechnology, Dendrimers are rolling in as a highly tempting class of drug delivery system for cancer therapy. Dendrimers are the best and smart choice as Nanocarriers to deliver one or more therapeutic agents safely and selectively to cancer cells. Dendrimers that have remarkable properties including membrane interaction, monodispersity, well-defined size, shape and molecular weight, etc. Functional groups that are present in the Dendrimers exterior also permit other chemical moieties that can actively target certain diseases which are now widely used as tumor-targeting strategies. There are three ways by which drugs interact with dendrimers, (a) physical encapsulation, (b) electrostatic interactions, and (c) covalent conjugations. This review represents the advantages of Dendrimers over conventional chemotherapy, toxicity, and its management. The anti-cancer drugs are delivered by using Dendrimers and recent advances in drug delivery by different types of Dendrimers.
Keywords
Dendrimer, cancer therapy, nanocarriers, drug delivery, diagnostic applications
INTRODUCTION
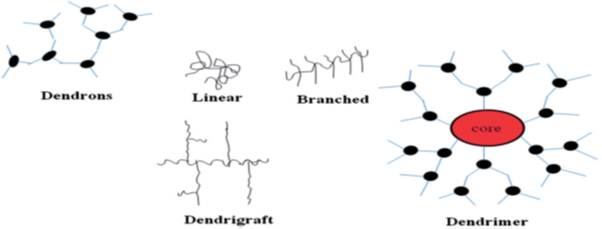
Cancer is one of the world's most nervous diseases with no significant cure for several types of tumors 1, 2. Cancer is a condition in which abnormal growth of the cell is going on with the potential to invade or spread to other parts of the body. The different challenges are facing an anti-cancer drug is–the first one is, a drug must be able to differences between a Cancer (transformed) cells from other healthy cells in the body. Secondly, they should provide adequately a high dose of a toxic agent to kill the cell. Furthermore, it has the property to successfully cures a patient by suppressing every cancer cell 3. About 40% of recently developed formulations are redundant to the pharmaceutical industry and may never help a patient because of poor bioavailability due to low water solubility or cell membrane permeability. The use of conventional formulations is limited by poor release patterns of drugs, poor solubility, and toxicity. New drug-delivery technologies based on nanomaterials may be a ray of hope to overcome these challenges 4. Nanotechnology is based on the materials having a size range in the dimensions of 1–100 nm. One of the most advanced nanomaterials, i.e., dendrimer, is being explored widely in biomedical applications due to well-defined size and shape 5. Dendrimers are highly branched spherical molecules having a well-defined chemical structure. They are defined as monodispersed, 3D, and highly branched polymers 6. In 1978, Fritz Vogtle and coworkers introduced dendrimer chemistry, and in 1985 Tomalia synthesized the first family of a dendrimer. Newcomers group reported the synthesis of dendrimer and named them arborols which is derived from the Latin word Arbor meaning ''tree''. A dendrimer is a highly branched synthetic polymer and consists of the monomer unit having a high degree of branching and hence known as the polymer of the twenty-first century 7. The structure of the dendrimers is shown in (Figure 1). There are three main components of dendrimer are central core, repetitive branching units, and terminal groups. Dendrimers have been successfully explored for the delivery of anticancer drugs as well as for theranostic applications in cancer therapy. Dendrimeric conjugates of an anticancer drug have shown the ability to bypass efflux transporter, to deliver the drug intracellularly, and to improve the bioavailability of loaded molecular cargo. Complexes of cisplatin with dendrimers have shown reduced cytotoxicity with significant anti-proliferative activity. Apart from the targeted delivery of chemotherapeutic agents, dendrimers are also being investigated for the delivery of diagnostic agents for targeted imaging of tumors 8, 9.
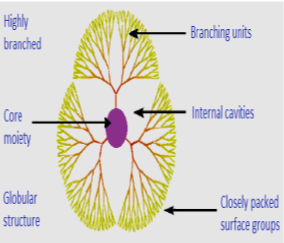
Advantages of Dendrimers over conventional anti-cancer agents
Dendrimers having appropriate Nano size ranging from 1-100 nm for pre detectable release profile, favorable pharmacokinetics, and targeting potentials 2, 10. They also can improve the solubility of poorly soluble anti-neoplastic drugs. due to the smaller size range of Dendrimers clearance rate is reduced through the reticuloendothelial system 11, 12. Multiple functional groups are present on the outer surface of Dendrimers, which can be used to attach vector devices for targeting to a particular site in the body. Numerous peripheral functional groups on Dendrimers is responsible for tumor cell-specific delivery.
Toxicity and its management
The toxicity is dependent on the dendrimer characteristics and can be related to the chemistry of the core but mainly to surface end groups 13, 14. Interaction of the cationic dendrimers surfaces with negative biological load membranes damaging cellular membranes causing hemolytic toxicity and cytotoxicity 15. This strong interaction with the negatively charged cell membranes can cause cell destabilization with leakage of cytoplasmic proteins and subsequently lysis 13, 16, 17. E. g.: Interaction with lipid bilayers of cells occurs with the cationic Dendrimer G7-PAMAM which comes to form holes 15-40 nm in diameter, which disturbs the flow of electrolyte causing cell death 15, 17, 18, 19. Surface modificationof dendrimers can be useful to improve their safety and can be easily achieved through conjugation molecules with the reactive terminal groups of nanocarriers (Table 1). PEG is frequently used to increase plasma circulation time and tumor accumulation through enhanced permeability and retention effect [EPR] 13, 20, 21, 22. Linking or conjugation of dendrimers with PEG chains has been shown as an important step in reducing the cytotoxicity of Dendrimers. PEGylation increase the physical dendrimer size which reduces renal clearance since the glomerular filtration limits reached 13, 23, 24, 25, 26.
|
S.N. |
Anti-cancer Drug |
Dendrimers type |
|
1. |
5-FU(5-Fluorouracil) |
PAMAM-mPEG-PDEA PAMAM Polyglycerol |
|
2. |
CDDP(Cisplatin) |
PAMAM PAMAM-COOH |
|
3. |
MTX(Methotrexate) |
Polyether-co-polyester PAMAM |
|
4. |
PTX(Paclitaxel) |
Poly Glycerol, PAMAM Triazine |
|
5. |
DOX(Doxorubicin) |
PEGylated-PAMAM, Polyester |
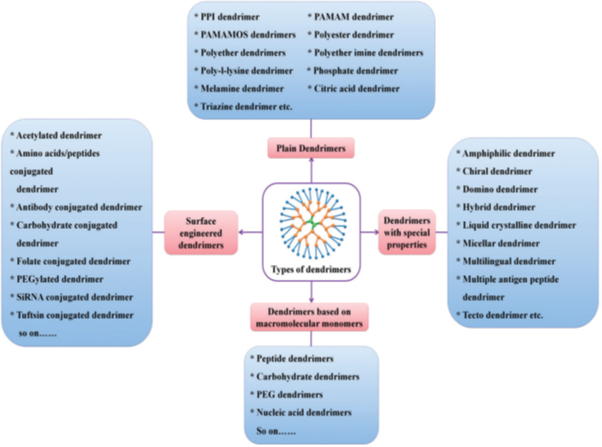
Terms and nomenclature in dendrimer chemistry:
Dendrimer chemistry as extra specialized research fields has its terms as described below:
Dendrigrafts
Dendrigraft polymers (Figure 2) are closest to Dendrimers structures. Functionalization and grafting reactions are used to synthesize the dendrigrafts. Dendrigrafts are defining as grafting linear chains onto a linear polymer substrate suitably functionalized with coupling sites that yield a comb-branched polymer structure 27.
Dendrons
Dendrons is the term used for a dendritic wedge without a core, the dendrimer can be prepared from assembling two or more dendrons. These dendrons (Figure 2) have been used
in the creation of huge numbers of dendrimers. Properties and characteristics of dendrimers depend upon the dendrons 28.
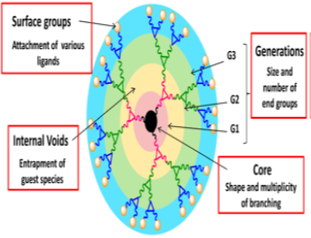
Generation
Dendrimers are synthesized by the repetition of a chemical reaction and grown out of the focal point, i.e. core. With each repetition, onelayer is formed around the core which is known as ‘generation number of dendrimers as shown in Figure 3 . The number of focal points when going from the core toward the dendrimer surface is known as generation number. Generation (Figure 3) is common for all dendrimer designs and the hyperbranched when going from the center of the dendrimer toward the periphery, resulting in homostructural layers between the focal points (branching points). Thus, a dendrimer having five concentric layers when going from the center to the periphery is denoted as the 5.0 G generation dendrimer 7, 16, 29.
End-group
End groups are also generally referred to as the 'terminal groups' or the 'surface groups' of dendrimers. The surface group is a slightly more accurate word, in the sense that the dendrimers branches can sometimes fold into the interior of the dendrimers. Dendrimers having amine end-groups are termed 'amino-terminated dendrimers' like polypropylene imine (PPI), polyamido amine (PAMAM), and poly-L-lysine (PLL) dendrimers 30, 31.
Methods for the synthesis of Dendrimers:
The following methods are generally used in the synthesis of Dendrimers-
a) Divergent growth method
b) Convergent growth method
c) Hyper-core and branched and method
d) Double exponential
a) Mixed growth methods
Divergent growth method:
In this method, the growth of dendrimers starts from the core and grows in an outward direction (Figure 4). This method was firstly introduced by Tomalia and Newkome and the name divergent came from the synthesis strategy in which dendrimers grow around the core. Divergent growth methods result in the sequential addition of generations to the core resulting in the 3D architecture of dendrimers with a simultaneous increase in the number of surface functional groups, molecular weight, and generations 7, 32.
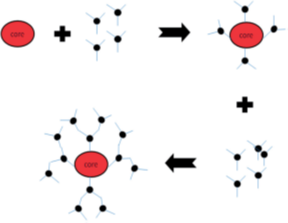
Convergent growth method
Mainly this method begins from the core and progresses toward inward (Figure 5). The branching units are grown and linked to one and other groups. When these branches are large enough, they are attached to the core molecule 7, 32.
Hypercore and branched method
Herein, pre-assembly of oligomers (polymers whose molecules consist of relatively few repeating units) join together to produce the desired structure of dendrimer in a few steps. This is the appropriate method overconvergent method because fewer steps are required for the synthesis of higher generations of dendrimer 7, 16.
Double exponential and mixed growth
In this method, dendrimers are synthesized by both divergent as well as convergent growth methods 7, 16. It is the most advanced method of dendrimer synthesis where both divergent and convergent methods are used to form a triangle called ‘Dendrimer’. The triangle may be used to repeat the growth process.
Dendrimers in anticancer drug delivery
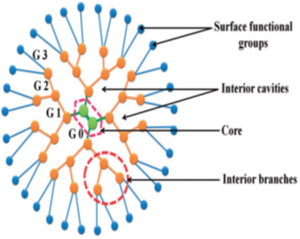
A dendrimer is a nanocarrier having a size of 1–100 nm. Dendrimers are promising candidates for improving the solubility and diminishing the toxicity of anticancer drugs as well as for targeted delivery to cancer cells and theranostic applications 4, 7 (Figure 6). Drug molecules may be covalently conjugated to the end groups of a dendrimer or entrapped inside the core via hydrogen bonding, hydrophobic linkage, or electrostatic interactions 33. The number of generations influences the drug loading capacity: a relatively high generation number provides more space for guest drugs and has a larger number of functional groups on the surface for drug conjugation. PPI, PAMAM, PLL, polypeptide, polyesters, polyether dendrimers, and dendrimers based on PEG, or carbohydrates have been mainly investigated for delivery anticancer drugs 31, 34. These Dendrimers have been investigated frequently as a drug carrier, due to the availability of high density of functional groups (amine groups) on the surface. Also, cationic charges of PPI, PAMAM, and PLL dendrimers enable the delivery of nucleic acids. Drug release from dendrimers depends on the type of interactions between a drug and a dendrimer 35. The anticancer drug interacts with dendrimers via three different mechanisms, i.e.
a) Physical encapsulation,
b) Electrostatic interaction,
c) Covalent conjugation
A summary of the mechanism of interaction of anticancer drugs with dendrimers is shown in (Table 2).
|
Sr. No. |
Drugs |
MOA |
Uses |
Drug dendrimer interaction |
Application |
|
1. |
5-Fluorouracil |
5-Fluorouracil is inhibited the formation of thymidylate from uracil |
Breast cancer Colorectal cancer |
Chemical conjugation |
Enhancement of solubility of 5-Fluorouracil |
|
2. |
Pancreatic cancer Cisplatin |
Attachment of alkyl groups to DNA bases, DNA damage via the formation of crosslinks |
Blader cancer Cervical cancer |
Covalent conjugation |
Improved loading efficiency, reduced cytotoxicity but significant anti-proliferative activity against lung cancercell |
|
3. |
Ovarian cancer Doxorubicin Hydrochloride |
Doxorubicin HCl inhibits topoisomerase II activity |
Gastric cancer Ovarian cancer Thyroid cancer Breast cancer |
Physical encapsulation |
Enhancement of solubility of Doxorubicin HCl |
|
4. |
Wilms tumor Methotrexate |
Inhibiting the metabolism of folic acid and enzymes in the synthesis of purines and pyrimidines required for cell proliferation |
Breast cancer Neck cancer Head cancer |
Physical encapsulation |
Improve bioavailability and toxicity |
|
5. |
Lung cancer Paclitaxel |
Promotes tubulin assembly, stabilize microtubules |
Breast cancer Lung cancer |
Physical encapsulation |
Enhanced water solubility compared to the pure drug |
|
6. |
Ovarian cancer Cisplatin |
Attachment of alkyl groups to DNA bases, DNA damage via the formation of cross-links |
Cervical cancer Ovarian cancer Bladder cancer |
Physical encapsulation |
Reduced toxicity |
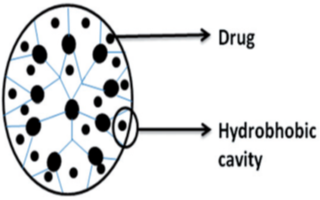
Physical encapsulation
Due to its spherical shape, unfilled internal cavities, and structural design, dendrimers can directly encapsulate guest molecules into the macromolecule interior (Figure 7). The empty internal cavities of dendrimers are hydrophobic in nature and interact with poorly soluble drugs through hydrophobic interactions 36. Nitrogen or oxygen atoms present in the internal cavities of dendrimers can interact with the drug molecules by hydrogen bond formation. The relationship between the internal cavities of dendrimers and drug molecules may involve some interactions like physical encapsulation, hydrophobic interaction, or hydrogen bonding.
Due to their inherent architecture, drug molecules can be loaded into dendrimers using their well-defined internal crevices by hydrophobic interactions or hydrogen bonding or by electrostatic interactions between drug ionic groups and oppositely charged dendrimer surfaces. The ability of Dendrimers to solubilize a variety of drugs including anticancer, anti-HIV, and anti-inflammatory agents has recently been reviewed 37. Compared with chemical conjugation, the physical incorporation of drugs has the advantage of straightforward, rapid preparation without adversely affecting drug pharmacological activity. Its disadvantages include low stability in terms of storage and premature drug release, batch-to-batch variation in concentration of the solubilized drug, and low-drug loading capacity 6 38.
Electrostatic interactions
A large number of amine groups and carboxyl groups on the surface of dendrimers have potential applications in enhancing the solubility of hydrophobic drugs by electrostatic interaction. Some drugs with carboxyl groups like ibuprofen, ketoprofen, diflunisal, naproxen, and indomethacin have been complexed with dendrimers by electrostatic interactions. Through electrostatic interactions, various ionizable drugs form complexes with the multifunctional surface of dendrimers having a large number of ionizable terminal surface groups. In PAMAM dendrimers, both primary amine and tertiary amine groups present on the surface and within the core, respectively, are titrable and have pKa values of 10.7 and 6.5, respectively. Hence, they possess the ionizable groups terminally as well as within their core and thus offer potential sites for drug interactions. Many of the drugs such as ibuprofen, piroxicam, indomethacin, and benzoic acid have been shown to form stable complexes through electrostatic interactions 39.
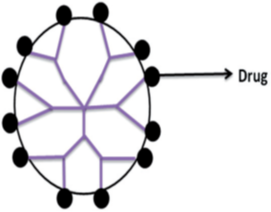
Covalent conjugation
The presence of large numbers of functional groups on the surface of dendrimers makes them suitable for the covalent conjugation of numerous drugs with relevant functional groups (Figure 8). In the case when a drug is covalently bound to dendrimers, its release occurs through chemical or enzymatic cleavage of hydrolytically labile bonds. Moreover, the drugs can be covalently conjugated to dendrimers through some spacers that may include PEG, p-aminobenzoic acid, p-amino hippuric acid, lauryl chains, etc., or biodegradable linkages such as amide or ester bonds. This prodrug approach has been found to increase the stability of drugs and has affected their release kinetics significantly.
Several researchers have successfully conjugated penicillin V, venlafaxine, 5-aminosalicylic acid, naproxen, propranolol with PAMAM dendrimers. The results have shown enhanced solubility and controlled release of drugs from these complexes in comparison to the plain drug. Apart from these, many anticancer drugs viz. cisplatin, doxorubicin, epirubicin, methotrexate, and paclitaxel have also been conjugated with Dendrimers and have shown potential drug targeting. For example, the epirubicin prodrug was developed by conjugating it with PEG dendrimers containing aminoadipic acid as branching molecules.
These conjugates exhibited increased blood residence time and showed improved therapeutic action. The study revealed that PEG dendrimers increased the stability of bound drug toward chemical degradation and hence can be usedpotentially in the development of prodrugs of large molecules 39.
Some of the drawbacks of physical drug incorporation canbe reduced by covalent linking of a drug to the dendrimer using a chemical approach which will also bring about selective drug release in vivo. Such an approach can generate structures with pre-measured drug content and enhanced stability. Drug release can be obtained by a change in the biological microenvironment such as variation in pH, temperature, or concentration of a specific enzyme 40.
Mechanism of drug delivery through Dendrimers
Due to compact and globular structure and a large number of surface functional groups, drug molecules can be encapsulated both in the interior of the dendrimers as well as attached or bound to the surface groups. Dendrimers can be used as drug carriers either by loading drugs within the dendritic structure or by interacting with drugs at their terminal functional groups through electrostatic or covalent bonds 10. The covalent bond is formed between the drug and exterior surface of dendrimers. The exterior surfaces of dendrimers have been reported as potential sites of interaction with drugs [44]. The number of drug molecules incorporated into dendrimers may depend on the structural design of dendrimers. The loading capacity is increased by the formation of a complex with the huge number of groups present on the surface of dendrimers. With the increasing generation of dendrimers, the number of groups present on the surface of dendrimers also increases 37.
There are two mechanisms for drug delivery-
-
In vivo degradation of a covalent bond between drugs dendrimer by suitable enzymes.
-
Releasing of the drug from dendrimer due to physical changes or stimulus like pH, temperature, etc.
Diagnostic applications of Dendrimers
Advances in nanotechnology have enabled the development of dendrimers that can be used as diagnostic aids in a diverse molecular imaging application, particularly diagnostic applications such as-
a) Cancer Imaging
b) Photodynamic therapy
c) Boron neutron capture therapy
d) To-dendrimer Nanodevice
e) Photothermal therapy
-
Cancer Imaging
-
Imaging techniques are used in cancer therapy to diagnose and identify the location of the disease sites, stage, plan treatment, and potentially find a recurrence.
-
MRI is a non-invasive technique for diagnosing tumors in soft tissues [46].
-
MRI agents were linked with dendrimer molecules for contrast enhancement, improved clearance characteristics, and potential targeting.
-
E. g.: Gadolinium contrast agents have been conjugated to PPI and evaluated for use as macromolecular MR contrast agents.
-
Computer tomography is a standard method of imaging associated with a cancer diagnosis [45].
-
-
Photodynamic therapy
-
This therapy depends on the activation of a photosensitizing agent with visible or near IR light.
-
When photosensitizer exposed to a specific wavelength of light, they produce a form of oxygen which kills cancer cells.
-
PDT has been shown to reduce tumors by direct cell killing, destruction of tumor neovasculature, and triggering of an acute inflammatory response that attracts leukocytes to the tumor [45].
-
-
Boron neutron capture therapy
-
BNCT is based on a lethal B(n,α) 7 Li capture reaction that occurs B is irradiated with low energy thermal neutrons to produce high energy α-particles and 7 Li nuclei
-
The emergence of BNCT as a significant clinical treatment has been limited by either lack of sufficient tumor-targeting or sub-therapeutic accumulation in malignant tissues.
-
To this end, macromolecular delivery vehicles have been prepared to enhance both the quantity of and targeting of B to tumor cells by conjugating Boron containing complexes to monoclonal antibodies or receptor targeting agents [46].
-
-
Tectodendrimer as nanodevice: Tecto dendrimers are multifunctional devices built from a cored dendrimer, surrounded by shell dendrimers. Each shell dendrimer performs one function
-
Photothermal therapy :
-
Gold-based nanoparticles have been developed that strongly absorb light in the near IR region, facilitating deep optical penetration into tissues, generating a localized lethal dose of heat at the site of the tumor.
-
Dendrimer-encapsulated gold nanoparticles have been prepared and identified for their potential use towards the photothermal treatment of malignant tissue.
-
E. g. Amine-terminated G5-PAMAM dendrimer-entrapped gold nanoparticles were prepared and covalently conjugated to fluorescein and folic acid for targeted delivery to tumor cells overexpressing folic acid receptors [46].
-
Conclusion
Dendrimer provides a unique platform for drug attachment that can bind and release drugs through several mechanisms. A dendrimer is a delivery vehicle for anticancer agents that are poorly soluble in water and can promote the transport of drugs across biological membranes.
Drug molecules can be encapsulated both in the interior of the dendrimers as well as attached or bound to the surface groups due to compact and globular structure and a large number of surface functional groups. Drugs can link with dendrimer through a strong covalent- and cleavable bond. Some problems occur in the case of drugs covalently linked to Dendrimers due to the formation of a stable bond. The degradation of the drug can be protected by the use of dendrimer carriers and inhibit renal clearance. Dendrimers have been used actively or passively to target a payload to particular destinations. Like any other nanomaterials, the possible biomedical or therapeutic applications of dendrimers would demand an unequivocal point of their safety which is a prerequisite for their regulatory approval. The known cidal activity of some dendrimers could be further exploited in the area of contraception.
ACKNOWLEDGMENT
I am Krishna Kumar immensely obliged and thankful to Director Amresh Gupta sir, Arpita Singh ma’am, Satyawan Singh sir & Swarnima Pandey ma’am of Goel Institute of Pharmacy & Sciences Lucknow for guiding me to write this review article.