Abstract
In this article, we discussed the type, preparation, and evaluation of liposomes. The term liposome means the body of lipid and it consisting of one or more phospholipid bilayers. liposomes useful in reproduction, reagent, and gear in various scientific regimentation, including mathematics and theoretical physics, chemistry, colloid science, biochemistry, biology, etc. these are one of the various drug delivery system used to target the drug to particular tissue because liposomes structure similar to lipid bilayer and cell membrane.
Keywords
Liposomes, preparation, evaluation
INTRODUCTION
Liposomes are small synthetic vesicles of spherical shape which can be developed from two major materials cholesterol and natural non-toxic phospholipids. Its hydrophobic and hydrophilic nature and are promising systems for drug delivery. In 1965, some researchers published the primary description of swollen phospholipid systems. Within a couple of years, a spread of enclosed phospholipid bilayer structures consisting of single bilayers, initially 'Bangosomes' then 'liposomes', were described. It has been expressed that phospholipids impulsively form closed structures when they are hydrated in aqueous solutions. Such vesicles that have one or more phospholipid bilayer membranes can transport aqueous or lipid drugs, counting on the character of these drugs. The lipid used to make liposomes belong to both hydrophilic and hydrophobic nature show (amphipathic nature) in aqueous media, their thermodynamic phase properties and self-assembling characteristics impression entropically focused attachment of their hydrophobic sections into spherical bilayers. Those layers are referred to as lamellae. Liposomes are used to improve the therapeutic index of both new and old drugs by modifying drug absorption, reducing metabolism, long biological half-life, or decrease toxicity. Drug distribution is then controlled primary properties of the carrier and not by the physicochemical character of the drug substance only. Lipids forming liposomes also maybe both types natural or synthetic, and liposome ingredients aren't particular of lipids, new generation liposomes also can be constituted from polymers. The composed liposomes may be natural or synthetic lipids or polymers, liposomes are biocompatible and biodegradable making them suitable for a different type of biomedical research. This feature, including biocompatibility and biodegradability both, makes it very useful, attractive as drug delivery vehicles 1, 2, 3, 4, 5, 6.
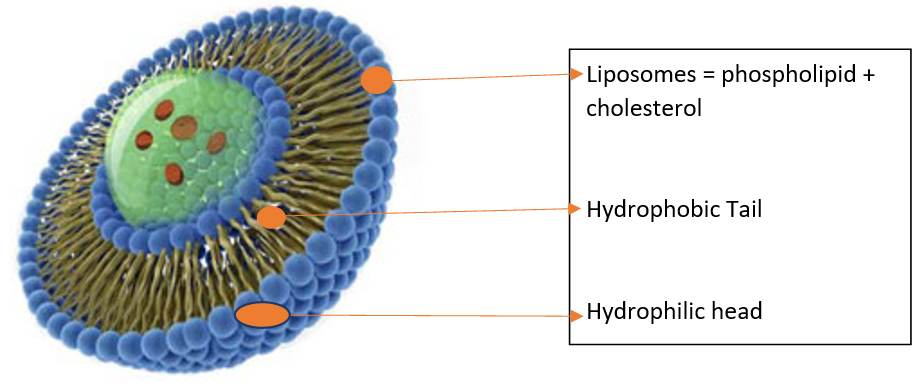
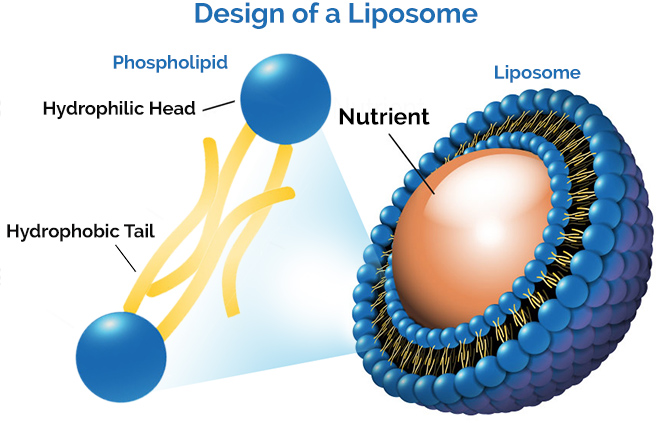
MECHANISM OF FORMATION OF LIPOSOME
A phospholipid is a backbone for the formulation of liposomes. A liposome may be a solution and core surrounded by a hydrophobic membrane, within the sort of a lipid bilayer, hydrophilic solutes dissolved within the core cannot readily pass through the bilayer. Hydrophobic substances are associated with the bilayer. To deliver the molecules to a site of action, the lipid bilayer can fuse with other bilayers like a cell membrane. This is a complex and non-spontaneous event. Liposomes are used as models for artificial cells. Liposomes can also be designed to deliver drugs in a different types of ways. Liposomes to implant low or high pH may be built such as dissolved aqueous drugs will be charged in the solution. The pH is naturally neutralized inside the liposome, the drug will be neutralized, make free it to freely undergo a membrane. These liposomes work to deliver drugs by diffusion instead of by direct cell fusion. A similar approach an often exploited within the biodetoxification of medicine by injecting empty liposomes with a transmembrane pH gradient. In this case, the vesicles act as sinks to scrub the drug in the blood circulation and prevent its toxic effect. Another strategy for liposome drug delivery is to focus on endocytosis events. These liposomes could also be digested while within the macrophage's phagosome, thus releasing its drug. Liposomes also fitted with opsonin and ligands to activate endocytosis into the other type of cells. The use of liposomes for the transformation or transfection of DNA into a number cell is known as liposome transfection. liposomes are often used as carriers for the delivery of dyes to textiles 7, 8, 9.
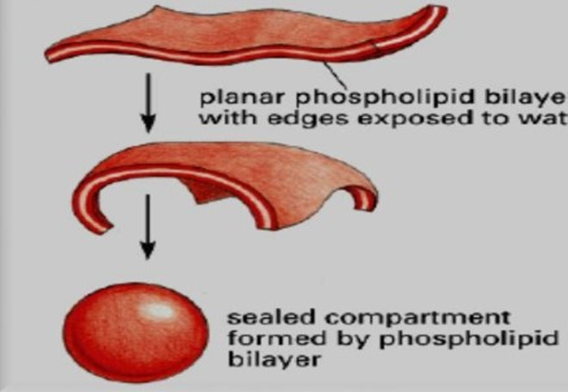
ADVANTAGES OF LIPOSOMES
-
Provide sustained release.
-
Nonionic
-
Liposomes are used as targeted drug delivery or site-specific drug delivery.
-
Stabilization of entrapped drug from a hostile environment.
-
Can carry both water and lipid-soluble drug.
-
The biodegradable drug can be stabilized from oxidation.
-
Improve protein stabilization.
-
Controlled hydration
-
Can be administered through various routes.
-
Alter pharmacokinetics and pharmacodynamics.
-
Liposomes proposed various advantages in delivering genes to cells.
-
Liposomes have both negatively and positively charged molecules.
-
Liposomes offer protection of DNA from degradative processes.
-
Liposomes may be carries large flinders of DNA, possibly as a chromosome.
-
Liposomes are targeted to specific cells or tissues.
-
Liposomes are a more effective and therapeutic index of drugs.
-
Liposomes help reduces the exposure of sensitive tissue to the toxic drug.
-
Springiness to couple with site-specific ligands to obtain active targeting 10, 11.
DISADVANTAGES OF LIPOSOMES
-
Production cost is high.
-
Leakage and fusion of encapsulated drug and/or molecules.
-
Sometimes phospholipid undergoes oxidation and hydrolysis-like reactions.
-
Short half-life.
-
Low solubility.
-
Fewer stables.
-
Quick uptake by cells R.E.S.
-
Batch to batch variation.
-
Difficult in large-scale manufacturing and sterilization.
-
Once administered, liposomes cannot be removed.
-
The allergic reaction may occur with liposomal constituents.
-
Possibility of dumping, due to faulty administration.
LIPOSOMAL DRUG DELIVERY SYSTEM
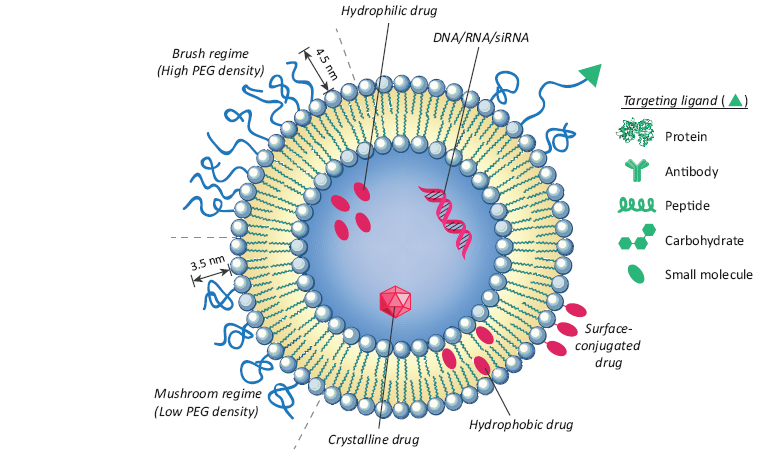
Structural and design considerations for liposomal drug delivery. Liposomal surface functionalized to provided deception through PEGylation promote receptor-mediated endocytosis by using targeting ligands like antibodies, peptides, proteins, carbohydrates, and different small molecules. PEGylation enlarges liposomal circulation half-life in vivo by decrease clearance, immune recognition, and the non-specific absorption of serum proteins. PEGylation density composes its structure at the liposome surface, with a density below 9-10% appropriate to a fungus-like circular structure and those above 9-10% adopting a more tough, extended, brush-like morphology. Diagnostics and chemotherapy may be encapsulated into the aquatic lumen, consolidated into the lipid bilayer, or conjugated to the liposome surface.
TYPES OF LIPOSOME
The liposomes have a size from very small (0.025 μm) to large (2.5 μm) vesicles. Moreover, liposomes may have one or bilayer membranes. The liposomal vesicle size is a rapid parameter in determining the circulation half-life of liposomes, and both size and number of bilayers affect the amount of drug encapsulation in the liposomes 12.
Based on their size and number of bilayers, liposomes can also be classified into two categories:
-
Multilamellar vesicles (MLV)
-
Unilamellar vesicles (ULV)
Unilamellar vesicles classified into two categories
-
Large unilamellar vesicles (LUV)
-
Small unilamellar vesicles (SUV)
METHODS OF PREPARATION OF LIPOSOMES 13, 14
General methods of preparation
-
Drying down lipids from organic solvent
-
Dispersing the lipid in aqueous media
-
Purifying the resultant liposome
-
Analyzing the final product
Methods of liposomes preparation and drug loading
-
Passive loading techniques
-
Active loading technique
Passive loading techniques include three different methods
-
Mechanical dispersion method
-
Solvent dispersion method
-
Detergent removal method (removal of non-encapsulated material)
Mechanical dispersion method
-
Sonication
-
French pressure cell: extrusion
-
Freeze-thawed liposomes
-
Lipid film hydration by handshaking, non-hand shaking, or freeze-drying
-
Micro-emulsification
-
Membrane extrusion
-
Dried reconstituted vesicles
EVALUATION OF LIPOSOMES
Liposomes are ready by various methods/techniques are to be evaluated for physical, chemical, and biological properties, as these influence the behavior of liposomes in vivo.
PHYSICAL PROPERTIES
Particle size
These can be determined by the following method.
a) Laser light scattering
b) Transmission electron microscopySurface charge
The passive, negative or natural charge on the surface of the liposomes is due to the composition is the head groups. The surface charge of liposomes governs the kinetics and extent of distribution in vivo, also as interaction with the target cells.The method involved within the measurement of surface charge is predicated on free-flow electrophoresis of MLVs. It utilizes a cellulose ester plate dipped in sodium borate buffer of pH 8.8. About moles of lipid samples are applied onto the plate, which is then subjected to electrophoresis at 4'C for 30 mins. The liposomes get bifurcated counting on their surface charge.Percent drug encapsulated
The quantity of drugs entrapped within the liposomes helps to estimate the behavior of the drug in a biological system. The %age of drug encapsulation is done by first separating the free drug friction from the encapsulated drug fraction.The encapsulated drug fraction is then made to lack the liposomes into an aqueous solution using suitable detergents.
The methods want to separate the free drug from the sample are:
a) Mini column centrifugation method
b) Protamine aggregates methodPhase behavior
At the transition, temperature liposomes undergo a reversible phase transition. The TC is that the indication of stability permeability and also indicates the region of drug entrapment. It is done by DSC.Drug release rate
The rate of drug release from the liposomes can be determined by in vivo assays which help to predict the pharmacokinetics and bioavailability of the drug. However, in vivo studies are found to be more complete. Liposomes encapsulating the tracer insulin are employed for the study. This insulin is preferred because it is released only within the ECF and undergoes rapid renal excretion of the face tracer coupled to the degradation rate constant o the tracer released from the liposomes.
CHEMICAL PROPERTIES
DETERMINATION OF PHOSPHOLIPID
The phospholipid content of liposomes an often determined directly by two assays, Bartlett assay, and steward assay.a) Bartlett assay
This method of determining the phospholipid is extremely sensitive and should produce erroneous leads to the presence of even trace amounts of phosphate. Therefore, borosilicate glass tubes and double–water is employed.
-
Initially the phosphorous present in the lipid bilayer of the sample is hydrolyzed to inorganic phosphate.
-
Then ammonium molybdate is added to convert inorganic phosphate to phosphomolybdic acid (PMP).
-
The sample is then treated with amino naphthyl sulphonic acid to quantitatively reduce the PMP to a blue-colored compound.
-
The intensity of the blue color produced can be measured by spectrophotometric means and the value is plotted on the standard curve to obtain the content of phospholipids.
b) Steward assay
This assay overcomes the drawbacks of Bartlett assay, but cannot be used to the mixture of unknown phospholipids.
-
The standard curve is prepared by the known concentration of phospholipids in chloroform with a 0.1 M solution of ammonium Ferro-thiocyanate reagent.
-
The sample is also treated with the same reagent and the optical density is determined at 485 nm.
-
The absorbance of the sample can be plotted on the standard curve to obtain the concentration of phospholipids.
CHOLESTEROL ANALYSIS
a) Qualitative analysis
Performed employing a capillary column crammed with fused silica.b) Quantitative analysis
The sample is reacted with a reagent and therefore the absorbance of purple color - complex is measured at 610 nm.
BIOLOGICAL CHARACTERIZATION
a) Sterility
The sterility test is performed by Aerobic or Anaerobic cultures.b) Pyrogenicity
The pyrogenicity test is performed by the Limulus amebocyte lysate (LAL) test.c) Animal toxicity
Animal toxicity test is performed by monitoring survival rate, histology, and pathology 15, 16.
APPLICATION OF LIPOSOMES
APPLICATIONS OF LIPOSOMES IN MEDICINES
Liposome encapsulation may change the changeable distribution of the encapsulated drug molecules inside the body, which can significantly reduce toxic side effects and increase the safety and efficacy of the treatment. Applications of liposomes in pharmacology and medicine are often divided into therapeutic and diagnostic applications of liposomes containing drugs or a spread of elements, and their utilization form reagent within the fundamental studies of cell interfaces, respect procedures, and of the mechanism of action of certain materials. The body protects itself with a posh defense system. Liposomes enter the body more amount of the drug cause thrombus formation and their surface is lastly inactivated by coating with biomacromolecules, while minor particles, also as microbes, bacteria and colloids are devoured by the cells of the system. This response of the system has triggered substantial efforts within the event of biocompatible and non-recognizable surfaces and has also, on the other hand, narrowed the spectrum of applications of microparticulate drug carriers only to the targeting of the precise same cells of the system. Although they are composed of natural substances they are quickly faraway from the circulation by the macrophages which are located mainly within the spleen, liver, and bone marrow 17, 18, 19, 20, 21.
LIPOSOMES IN INFECTIOUS AND PARASITIC DISEASES
Liposomes within the body digested by phagocytic cells in case of intravenous administration, they're ideal vehicles for the targeting of drug molecules into these macrophages. The most important examples of the 'Trojan horse-like' mechanism are various parasitic diseases which normally consist of the cells of the mononuclear phagocytic system. Liposomes accumulate within the same cell population which is infected and thus offer a perfect drug delivery vehicle. Similar approaches are often implemented in antibacterial and antiviral therapy. Several other routes, like a topical application or pulmonary (by inhalation) administration, also are being considered. Automatic targeting of liposomes to macrophages is often exploited in several other ways, including by macrophage activation and injection. Some general toxins persuade rigid macrophage response which results in macrophage activation. Normally, this is often done by administration of alum or killed bacteria, and liposomes offer a chic alternative. Practically, liposomes are getting used in animal vaccination already since 1988 and this is the more effective and safer 22, 23, 24, 25, 26.
LIPOSOMES IN ANTICANCER THERAPY
Recent studies commonly showed less toxic effects of the liposomal encapsulated drug, but in most of the cases, the drug molecules were not bioavailable, resulting not only in reduced toxicity but also in greatly compromised efficacy. hapless, this was also found to be true for the type of primary and secondary liver tumors. Several different types of liposome formulations of various anticancer agents were shown to be less toxic than the free drug. It mostly used in humans generally showed reduced toxicity and better tolerability of drug administration but with not too much hope of importance. Promiscuous apart formulations are in several stages of clinical trials and show mixed results 27, 28.
LIPOSOMES FOR RESPIRATORY DRUG DELIVERY SYSTEM
Liposomes are widely used in various types of respiratory disorders. Liposome in the form of aerosol has several advantages over ordinary aerosol, like – 29.
-
Sustained release
-
Prevention of local irritation
-
Reduced toxicity
-
Improved stability in the large aqueous core
Many types of injectable liposomal products are now in the market including Ambisome, Fungisome, and mycoses. To happen efficient, liposomal drug delivery structure for the lungs is dependent on the following parameters:
-
Lipid composition
-
Size
-
Charge
-
Drug and Lipid ratio
-
Method of delivery
LIPOSOMES OF BRAIN TARGETED DRUG DELIVERY
Yagi et al. have developed a liposome that can pass through the blood-brain barrier (BBB) and reach the human glioma. This deputed sulfatide and monoclonal antibodies as the sensory device to increase the targeting ability of the liposome. Egg phosphatidylcholine (contain both saturated and unsaturated lipids) liposomes coated with CHP also were valuable collected in brain tumors of the rat. The tissue distribution of the liposome with/without CHP-coating was investigated. Distribution of the CHP-coated liposome increased by 4.5 times within the tumor and by 2.1 times within the ipsilateral brain and decreased by 4 times within the spleen compared with that of the control liposome. The survival of drug implanted rats was investigated by the utilization of liposomes during which an antitumor drug was loaded. The runs, of cisplatin (CDDP), were administered via the specific route into rats, 5 days after tumor inoculation. The average survival was 35 days for the group treated with the CHP-coated liposomes. This was statistically significant compared with the case of the untreated group 30, 31.
OTHER APPLICATIONS
-
Liposomes are used for the treatment of heavy metal poisoning (Chelation therapy).
-
Liposomes as drug/protein delivery vehicle.
-
Liposomes act as a carrier for the drug in vivo and invitro.
-
In the targeted drug delivery system liposomes are used as anticancer drugs such as methotrexate, actinomycin-D.
-
Liposomes are used as a pulmonary drug delivery system (with the help of inhalational devices like nebulizers are used to produce an aerosol of droplets containing liposomes).
-
Liposomes for topical application (drugs like triamcinolone, methotrexate, benzocaine, corticosteroids, etc. can be successfully incorporated as topical liposomes).
-
Used in recombinant DNA technology.
-
The most important use of liposomes in cell biology is to manipulate the status of membrane lipid.
-
Sustained or controlled release drug delivery system.
-
Liposomes in antimicrobial, antifungal, and antiviral therapy.
-
Liposomes as protein carriers in immunology.
-
Intracellular drug delivery system
-
Site-avoidance delivery
-
Enzyme replacement
-
Oral drug delivery
-
In Tumors therapy
-
In Gene therapy
-
Formulation aid
-
In immunology
-
Cosmetics
CONCLUSION
Liposomes have been recognized as extremely useful carrier systems for targeted drug delivery. Liposomes are used in broad areas of pharmaceutical applications. It showing particular promise as an intracellular delivery system for antisense molecules, DNA, and also protein and peptides. It also promotes targeting the particular disease cells within the disease site. Finally, liposomal drugs exhibits reduce toxicities. And its enhanced drug delivery to disease location along with long circulation residence time, are now achieving clinical acceptance. Many liposomal products are already in the market and many are under clinical trial to get approval.