Fungi associated with leaf spots of sugarcane (Saccharum officinarum L)
Abstract
Survey for fungi associated with leaf spots of sugarcane in Chittoor and Nellore districts of Andhra Pradesh during 1986-87 revealed that 26 species of fungi belonging to 10 genera were found associated with leaf spots of sugarcane. The leaf spots found were minor ones and their incidence was meagre ranging from 1 to 3 per cent in both the districts. In common, the incidence was more in ratoon crop and also in the plant crop aged more than 6 months. The different types of minor leaf spots found on sugarcane in the 2 districts of Chittoor and Nellore were described as : Spindle-shaped spots; dark brown linear blotches; reddish brown spindles; reddish brown lesions; Phyllachora leaf stripes; dark brown elongated lesions; dark purple streaks; dark purple midrib streaks; dark red linear lesions and dark red midribs. The morphology and taxonomy of the fungi isolated from the leaf spots were studied and the fungi were identified as belonging to 26 species classifiable into 10 genera
Keywords
Sugar Cane, Saccharum officinarum L., Leaf Spot, Ascomycetes, Coelomycetes, Hyphomycetes, Photomicrography, Pathak, Gohnston
Introduction
Sugarcane (Saccharum officinarum L.) holds a prominent position in India as the main source of sugar and as a commercial crop of economic importance. It occupies an extent of 28.62 lakh hectares in the country producing 1716.81 lakh tonnes of cane and 171.681 lakh tonnes of sugar. Andhra Pradesh occupies fourth position in area with 1.36 lakh hectares and fifth position in sugar production with 9.575 lakh tones 1. Wide fluctuations in area, from 1.14 to 1.95 lakh hectares, and cane production, from 84 to 128 lakh tonnes occurred in Andhra Pradesh during the last decade. Dowing to several factors such as weather, pest and disease incidence 2. Of these natural factors, the disease problems are of the greatest concern to the cane growers and sugar industry. 2 listed 36 diseases on sugarcane in Andhra Pradesh. Of these, 13 are leaf spots caused by fungi. 3 listed 11 fungal leaf spots on sugarcane in India. 4 listed 26 fungal Leaf spots on sugarcane in the world.
Recently a few new fungal pathogens have been reported, inciting leaf spots on sugarcane in India and abroad. Leaf scorch was caused by a new species of Leptosphaeria viz., L. bicolor in Kenya 5. Leaf scorch caused by Staqonospora sacchari was recorded for the first time in India 6. 7 reported a new leaf spot disease, 'Long Brown Stripe' caused by Exserohilum halodes from Andhra Pradesh. He also reported that 50-55 per cent of the total green leaf of the plant was affected by the pathogen and there was a depression of about 25 per cent in juice sucrose in cane stalks with partially dried foliage due to the disease.
In Chittoor and Nellore districts of Andhra Pradesh many new sugarcane varieties are being cultivated. Sufficient research work has not been done in these two districts to enlighten the fungal leaf spot diseases on sugarcane. Hence, detailed investigations were undertaken to survey for the incidence of fungal leaf spot diseases of sugarcane in Chittoor and Nellore districts. The results of the investigation on the survey isolation, morphological study and identification of the fungi associated with leaf spots of sugarcane are presented in this dissertation.
Materials and Methods
2.1 DISEASE SURVEY
Plant and ratoon crops of sugarcane varieties grown in the fields of the farmers of Chittoor and Nellore districts, Agricultural Research Station of the Andhra Pradesh Agricultural University (A.P.A.U) at Perumallapalle and the Sri Venkateswara Agricultural College Farm (A.P.A.U) at Tirupati in Chittoor district were surveyed for the incidence of leaf spot diseases during 1986-87. For recording the incidence, the test plot was divided at random into 10 different spots. In each spot, 10 leaves were randomly selected and observed for the leaf/spots. The disease incidence was calculated by the following formula:
The average of 10 such observations was recorded in each plot.
2.2 COLLECTION OF DISEASED LEAVES
Diseased leaves from plant and ratoon crops of different varieties of sugarcane were collected in polythene bags and brought to the laboratory.
2.3 EXAMINATION OF SPECIMENS
The leaf spot symptoms were described while the leaves were fresh and green. The specimens were labelled and preserved in paper bags for further studies. The leaf spots were critically examined under stereo binocular microscope for the presence of any fungal fructifications and other sporulating structures.
2.4 LABORATORY TECHNIQUES
The general laboratory techniques followed were those detailed by Pathak (1974) and Johnston and Booth (1983) with modifications wherever necessary for the preparation of media, isolation of single spores and for the maintenance of cultures.
2.5 ISOLATION OF FUNGI ASSOCIATED WITH LEAF SPOTS
The leaves showing typical symptoms of fungal leaf spots were cut into bits with the spot surrounded by a little healthy tissue. Such bits were surface sterilized in 0.1 per cent mercuric chloride solution for 1 minute followed by thorough washing with sterile water. Some of the surface sterilized bits were kept on moist blotters placed in petri plates and some were placed on the surface of PDA and OMA slants in test tubes at the rate of one bit per slant and incubated at room temperature (28-32 °C) for one week. The fructifications or the individual fungal colonies developed on the leaf spots plated on moist blotters were examined under stereo binocular microscope from 3rd day onwards and transferred to petri plates and slants containing PDA/OMA medium, under aseptic conditions by using a sterile inoculation needle dipped in sterile water. The fungi were brought into pure culture by single spore isolation and maintained by sub-culturing on PDA/OMA slants for further studies.
2.6 OBSERVATION OF FUNGAL COLONIES
Regular observations of fungal colonies that appeared on the leaf spots were made under stereo binocular microscope from 3rd day onwards.
2.7 MORPHOLOGICAL AND TAXONOMIC STUDY OF THE FUNGI
Colonies of particular fungus exhibiting the slightest variation in their characters were critically examined under stereo binocular microscope. Temporary aqueous mounts were prepared by picking up the individual fungi or fructifications observed on different leaf spots. Semipermanent mounts were prepared with lactophenol and the mounts were sealed with DPX mountant. The species were identified based on the morphological features and dimensions of the various structures recorded by examining under compound research microscope at 8 x 10; 8 x 45 and 8 x 100 magnifications. For identifying the fungi relevant monographs and other literature were referred to.
2.8 PHOTOMICROGRAPHY
Conidiophores, conidia, pycnidia and perithecia were mounted in water and photomicrographs were taken at 7 x 45 magnifications.
EXPERIMENTAL RESULTS
3.1 ASSESSMENT OF LEAF SPOTS
In both the districts of Chittoor and Nellore where the survey for leaf spot diseases on sugarcane was conducted, the incidence of well recognised leaf spots like ring spot, yellow spot, eye spot and brown spot was practically not observed on any of the varieties. However, certain other leaf spots could be found to an extent of 1 to 3 per cent on CoT. 8201; 1 to 2 per cent each on Co. 62175 and on Co. 7219; 2 per cent on Co. 6907; 1 percent each on Co. 8014, Co. 8013 and Co. 997 varieties grown in the farmers' holdings in Chittoor district. Two per cent infection of leaf spots of similar nature was observed on each of the varieties Co. 7219, CoT. 8201 and Co. 62175 grown in the Agricultural Research Station, Perumallapalle of Chittoor district/ whereas the leaf spot infection was 1 per cent each on the varieties Co. 419, CoA. 7602, and Co. 62175 grown in the Sri Venkateswara Agricultural College Farm, Tirupati of Chittoor district.
In respect of Nellore district, the types of leaf spots were found to an extent of 1 to 3 per cent on CoT. 8201, 3 per cent on Co. 62175 and 1 to 2 per cent each on Co. 7219, CoC. 776 and Co. 6907 varieties grown in the farmers' holdings.
In both the districts, the incidence was more in the ratoon crop and also in the plant crop aged more than 6 months,
3.2 DESCRIPTION OF THE LEAF SPOTS
The leaf spots occurring on different sugarcane varieties are described separately.
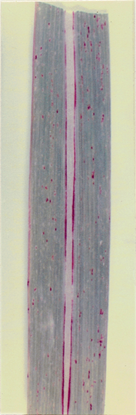
4.2.1 Spindle-shaped spots (Fig. 1)
4.2.2 Dark brown linear blotches (Fig. 2a)
4.2.3 Reddish brown spindles (Fig. 2b)
4.2.4 Reddish brown lesions (Fig. 3)


4.2.5 Phyllachora leaf stripes (Fig. 4)


4.2.6 Dark brown elongated lesions (Fig. 5)
4.2.7 Dark purple streaks (Fig. 6a)


4.2.8 Dark purple midrib streaks (Fig. 6b)
4.2.9 Dark red linear lesions (Fig. 7)
4.2.10 Dark red midribs (Fig. 8)

3.3 SYSTEMATIC ACCOUNT OF THE DIFFERENT LEAF SPOT FUNGI ISOLATED
A brief morphological and taxonomic account of fungi isolated from different leaf spots on sugarcane in Chittoor and Nellore districts of Andhra Pradesh is given:
The different fungi isolated belonged to 10 genera comprising of 26 species. Of these 2 genera belonged to Ascomycetes, 1 genus to Coelomycetes and the other 23 species representing 7 genera were classified under Hyphomycetes. All the 26 species were identified and described.
List of the fungi isolated
I. ASCOMYCETES: 1. Chaetomium sp.2. Phyllachora sp.
II. COELOMYCETES: 3. Phoma sorghina
III. HYPHOMYCETES: 4. Alternaria alternata 5. A. phragmospora 6. Cladosporium oxysporum
7. Curvularia borreriae 8. C. brachyspora 9. C. clavata 10. C. lunata 11. C. lunata var. aeria 12. C. pallescens
13. C. robusta 14. C. tuberculata 15. Drechslera australiensis 16. D. halodes 17. D. hawaiiensis 18. D. holmii
19. D. longirostrata 20. D. rostrata 21. D. sacchari 22. D. spicifer 23. D. stenospila 24. Myrothecium roridum
25. Fusarium sp. 26. Nigrospora sp.
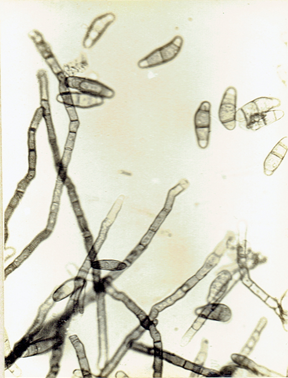
Phoma sorghina (Sacc.) Boerema et al. 1973. Persoonia 7: 134.(Fig. 9)
Alternaria alternata (Fr.) Keissler, 1912. Beih. Bot. Zbt.
Alternaria phragmospora Van Emden, 1970, Acta bot. neerl.
19: 393-400. Ellis, M.B. 1971. More Dematiaceous Hyphomycetes. 507 pp.(Fig. 11)
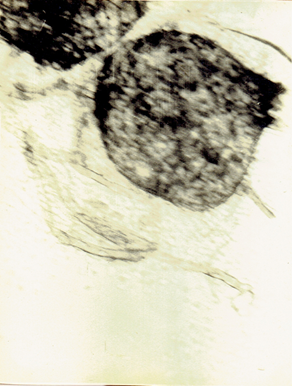
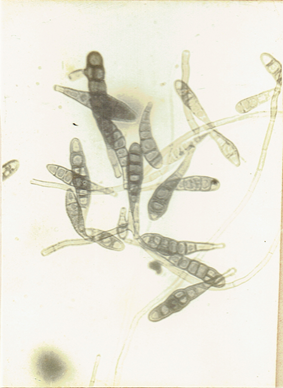
Chaetomium sp. Ames, L.M. 1969. A monograph of the Chaetomiaceae, 66 pp.(Fig. 12)
Cladosporium oxysporum Bark & Curt., 1868, J. Linn. Soc.
10: 362. Ellis, M.B., 1971. Dematiaceous Hyphomycetes, 608 pp.(Fig. 13)
Curvularia brachyspora Boedijn, 1933. Bull. Jard. Bot.
Buitenz, Ser. 3, 13 : 120-134. Ellis, M.B. 1966. Mycol. pap. 106: 2-43. Ellis, M.B. 1971. Dematiaceous Hyphomycetes. 608 pp, (Fig. 14)
Curvularia borreriae (Viegas) M.B. Ellis. Ellis, M.B. 1971. Dematiaceous Hyphomycetes, 608 pp.(Fig. 15)
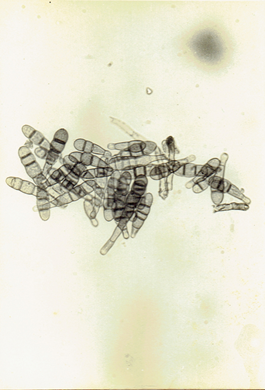
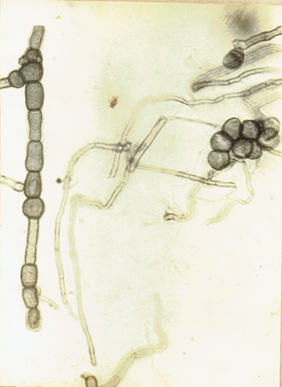
Fig.11b

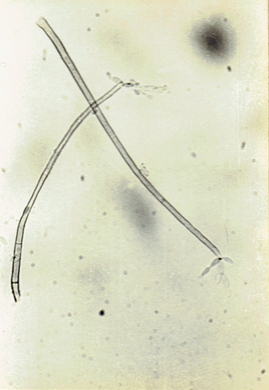
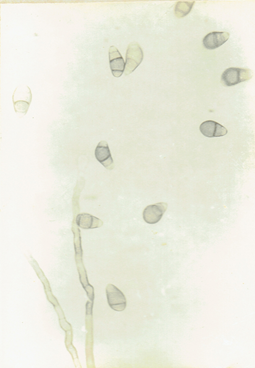
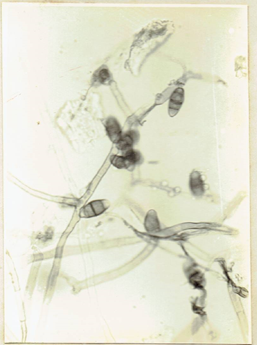
Curvularia clavata Jain. 1962. Trans. Brit. Mycol.
Soc. 45 : 542. Ellis, M.B. 1971. Dematiaceous Hyphomycetes. 608 pp.(Fig. 16)
Curvularia lunata var. aeria (Batista, lima and Vasconceus)
Ellis, M.B. 1966. Mycol. pap. 106: 2-43.
Ellis, M.B. 1977. Dematiaceous Hyphomycetes, 608 pp.(Fig. 17)
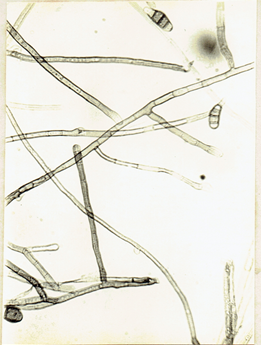

Fig. 16b
Curvularia lunata (Wakker) Boedijn, 1933. Bull. Jard. bot. buitenz. Ser. 3, 13: 127. Subramanian, C.V. 1971. Hyphomycetes. 930pp. Ellis, M.B. 1971. Dematiaceous Hyphomycetes. 608 pp.(Fig. 18)
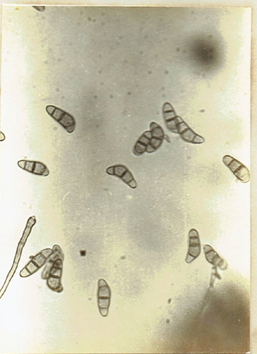
.Curvularia pallescens Boedijn. 1933. Bull. Jard bot. Buitenz. Ser. 3, 13: 127-8. Subramanian, C.V. 1971. Hyphomycetes. 930pp. Ellis, M.B. 1971. Dematiaceous
Hyphomycetes. 608 pp. (Fig. 19) Curvularia robusta Kilpatrick and Luttrell, 1967.
Mycologia 59: 888-892. Ellis, M.B. 1971. Dematiaceous Hyphomycetes. 608 pp.(Fig. 20)
Curvularia tuberculata Jain, 1962. Trans. Brit, mycol.
Soc. 45: 539-544. Ellis, M.B. 1971. Dematiaceous Hyphomycetes. 608 pp.(Fig. 21)

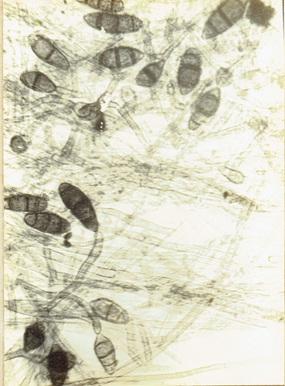
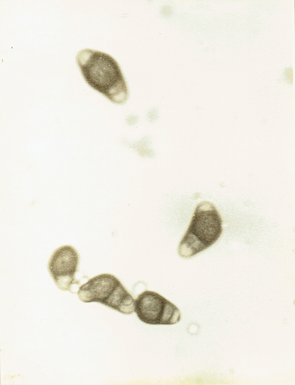
Drechslera halodes (Drechsler) Subram. and Jain, 1966.
Curr. Sci. 35: 354. Subramanian, C.V. 1971. Hyphomycetes. 930pp. Ellis, M.B. 1971. Dematiaceous Hyphomycetes. 608 pp.(Fig. 22)
Drechslera australiensis (Bugnicourt) Subram. and Jain ex M.B. Ellis, Subram. and Jain, 1966, Curr. Sci., 35: 354.
Ellis, M.B. 1971. Dematiaceous Hyphomycetes, 608 pp.-(Fig. 23)
Drechslera hawaiiensis (Bugnicourt) Subram. and Jain ex.
M.B. Ellis, Subram. & Jain, 1966. Curr. Sci. 35:
354. Ellis, M.B. 1971. Dematiaceous Hyphomycetes, 608 pp.(Fig. 24)
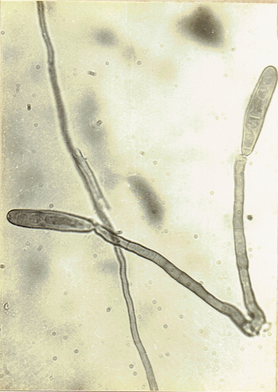
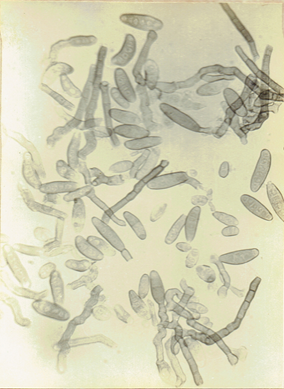
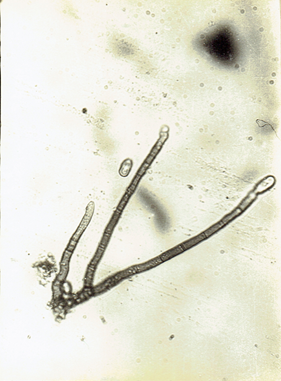
Fig.23 b
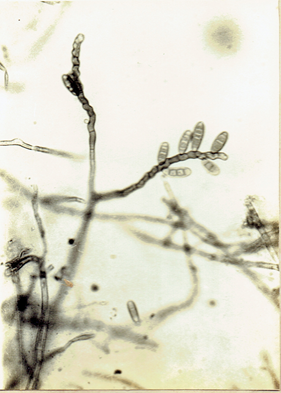

Drechslera state of Trichometasphaeria holmii Luttrell/
1963. Phytopathology, 53: 281-285. D. holmii (Luttrell) Subram. & Jain, 1966. Curr. Sci.35:354. Ellis, M.B. 1971. Dematiaceous Hyphomycetes. 608 pp. (Fig. 25)
Drechslera rostrata (Drechsler) Richardson and Fraser, 1968.
Trans. Brit. Mycol. Soc. 51 : 148. Subramanian, C.V. 1971. Hyphomycetes. 930pp. Ellis, M.B, 1971. Dematiaceous Hyphomycetes. 608 pp. (Fig. 26)
Drechslera longirostrata (Subram.) Subram., 1970. Proc.
First International symposium on Plant Pathology,
New Delhi: 194-203. Subramanian, C.V. 1971. Hyphomycetes. 930pp. (Fig. 27)
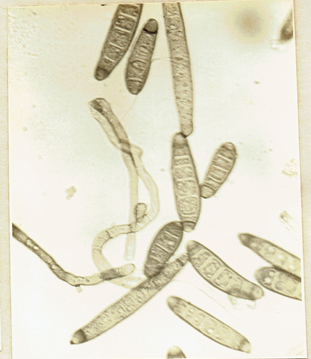
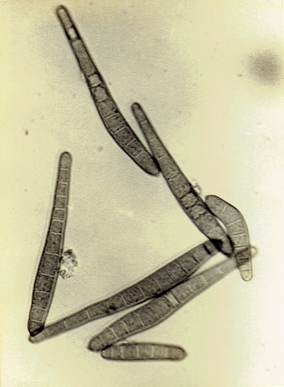
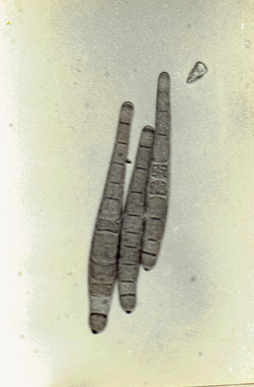
Drechslera state of Cochliobolus spicifer Nelson, 1964.
Mycologia 56: 198. Subramanian, C.V. 1971. Hyphomycetes. 930 pp. Ellis, M.B. 1971. Dematiaceous Hyphomycetes. 608 pp.(Fig. 28)
Drechslera stenospila (Drechsler) Subram. & Jain, 1966.
Curr. Sci. 35: 354. Ellis, M.B. 1976. More Dematiaceous Hyphomycetes. 508 pp. (Fig. 29)
Drechslera sacchari (Butler) Subram. & Jain, 1966.
Curr. Sci. 35: 354. Ellis, M.B. 1971. Damatiaceous Hyphomycetes. 608 pp. (Fig. 30)
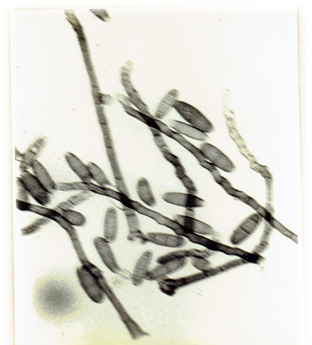

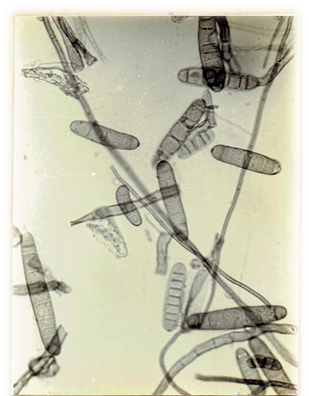

Myrothecium roridum. Tode ex Fr. 1829, Syst. mycol. 3: 217. Ellis, M.B. 1971. Dematiaceous Hyphomycetes. 608 pp. (Fig. 31)
Fusarium sp. Booth, C. 1971. The genus Fusarium. 237 µm. (Fig. 32)
Niqrospora sp. Ellis, M.B. 1971. Dematiaceous Hyphomycetes, 608 pp. (Fig. 33)
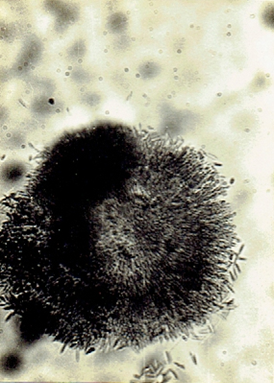
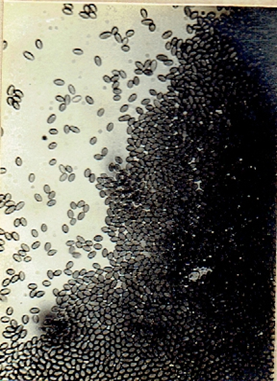
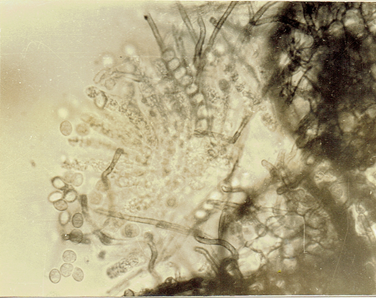
Phvllachora sp.
Phyllachora NKe. Fkl. Symb; Myc. 216: 186 9;
Cf. Petr. Ann. Myc. 22:1 1924; 25: 328, 1927. Frederic E. Clements and Cornelius L. Shear, 1973.
The Genera of Fungi. 496 pp. (Fig. 34)
DISCUSSION AND CONCLUSIONS
The present studies include survey for the incidence of leaf spots on sugarcane varieties cultivated in Chittoor and Nellore districts of Andhra Pradesh/ study of symptoms, isolation/ morphological studies and identification of the fungi associated with leaf spots.
During the survey, the occurrence of well recognised leaf spots such as ring spot, yellow spot, brown spot and eye spot was not observed. However, the incidence of certain other types of minor leaf spots could be recorded on sugarcane varieties cultivated in these areas.
In Chittoor district the percentage of incidence of fungal leaf spots ranged from 1 to 3 in the sugarcane varieties CoT. 8201, Co. 62175, Co. 7219, Co. 419, CoA. 7602, Co. 997, Co. 8013 and Co. 8014 cultivated in farmers' fields. The incidence was 1 per cent in the varieties Co. 419, CoA.7602 and Co. 62175 grown in the Sri Venkateswara Agricultural College Farm, Tirupati, whereas it was 2 per cent in the varieties CoT. 8201, Co. 7219 and Co. 62175 raised in the Agricultural Research Station, Perumallapalle. The incidence of leaf spots recorded varied from 2 to 3 per cent in the
varieties CoT. 8201, Co. 62175, Co. 6907, CoC. 776 and Co. 7219 grown in the farmers' holdings in Nellore district.
The leaf spots observed on sugarcane varieties of these areas were not considered to be of any serious concern to the growers as they were only minor in nature and their per cent incidence recorded was very low.
In the present survey, well recognised leaf spots such as ring spot, yellow spot, brown spot and eye spot which are generally found in low percentages in the coastal areas of Andhra Pradesh were not observed on sugarcane varieties cultivated in these areas. This is probably due to the dry weather prevailing in most part of the year; and even in the North East monsoon rainy season (October-December), the humidity per cent varies from 52 to 78 in Chittoor district (Appendix IV), compared to the coastal districts where the humidity per cent remains higher and favourable for more number of days during the rainy season for the development of leaf spot diseases.
While searching for the leaf spots on sugarcane, care was taken to eliminate the necrotic patches appeared to have been caused by the attack of insects and mites.
The various types of sugarcane leaf spots described in the present work viz., spindle-shaped spots (Fig. 1), dark brown linear blotches (Fig. 2a), reddish brown spindles (Fig. 2b), reddish brown lesions (Fig.3), Phyllachora leaf stripes (Fig. 4)/ dark brown elongated lesions (Fig. 5), dark purple streaks (Fig. 6a), dark purple midrib streaks (Fig. 6b) dark red linear lesions (Fig.7) and dark red midribs (Fig. 8) are considered minor, based on the nature and extent of damage caused by them.
All the fungi isolated from the leaf spots of sugarcane were described and identified. The following fungi were found associated with the leaf spots studied here* Chaetomium sp., Phoma sorghina, Alternaria alternata, A. phragmospora, Cladosporium oxysporum, Curvularia brachyspora, C. borreriae, C. clavata, C. lunata, C. lunata var. aeria, C. pallescens, C. robusta, C. tuberculata, Drechslera australiensis, D. halodes, D. hawaiiensis, D. holmii, D. longirostrata, D. rostrata, D. sacchari, D. spicifer, D. stenospila, Fusarium sp., Myrothecium roridum, Nigrospora sp., and Phyllachora sp.
The characteristic spindle-shaped leaf spots described on the sugarcane variety CoT. 8201 collected from Chittoor district were constantly found associated with Curvularia tuberculata. Leaf spotting associated with C. tuberculata was not recorded on sugarcane and it appears to be the first record on sugarcane.
Dark brown linear blotches described on the sugarcane variety Co. 62175, grown in Chittoor district was found associated with Alternaria alternata, Nigrospora sp. and Drechslera holmii.
Nigrospora sacchari was isolated from sugarcane in Mauritius 8. Colonization of N. sphaerica and Alternaria tenuis on ageing leaves of B. 3439 variety of sugarcane from Jamaica was reported 9. Drechslera holmii associated with leaf spots of sugarcane forms a new record in the present work.
Reddish brown spindles described on the sugarcane variety Co. 6907, collected from Chittoor district was found associated with Curvularia pallescens, Drechslera halodes and D. spicifer.
Curvularia pallescens associated with leaf spots of sugarcane forms a new record as it was not reported in the literature. Association of Drechslera (Helminthosporium) halodes with dark brown stripes on the sugarcane seedlings was reported from Sugarcane Breeding Station, Coimbatore/
India 10. The same fungus was also reported from India, causing severe sugarcane leaf blight 11. Drechslera spicifer isolated from the leaf spot in the present work was reported as the causal agent of seedling blight of sugarcane in India 12.
Colonies of Curvularia lunata, Alternaria alternata and Drechslera stenospila could be isolated from reddish brown lesions described on the sugarcane leaves of Co. 8014 collected from Chittoor district.
Curvularia lunata was found to be pathogenic on sugarcane, causing seedling blight [14 &15]. Same fungus was found on the ageing leaves of sugarcane from Jamaica 9. Sugarcane leaf spots due to C. lunata were given in the appendix of sugarcane diseases recorded in different parts of the world 4. Curvularia leaf spot caused by C. lunata was observed in Andhra Pradesh 2. Drechslera stenospila isolated along with the above 2 fungi from reddish brown lesions in the present work was listed as the causal agent of brown stripe of sugarcane 2, 3, 4. Typical Brown stripe attributed to D. stenospila was not observed in the present investigation.
Phyllachora leaf stripes containing black perithecia of Phyllachora sp., Curvularia brachyspora and a species of Fusarium were described on the sugarcane variety Co. 7219, cultivated in Chittoor district. Phyllachora sacchari causing leaf spots on sugarcane was reported from Philippines 13. Leaf spot due to this fungus was reported from India 4. Fusarium sp. associated with red line disease of sugarcane was observed in Andhra Pradesh 2.
Drechslera sacchari along with Curvularia borreriae, C. robusta and Cladosporium oxysporum were isolated from dark brown elongated lesions formed on the sugarcane variety CoA. 7602, collected from Chittoor district.
D. sacchari is known in the literature as the causal organism of eye spot on sugarcane 2, 3, 4. However, typical eye spot symptoms were not observed in the present survey and this fungus was found associated with other organisms in the dark brown elongated lesions. The Curvularia species viz., C. borreriae and C. robusta appear to be the first records on sugarcane.
Dark purple streaks associated with Myrothecium roridum were observed on sugarcane variety CoT. 8201, collected from Nellore district. The fungus is known to cause sheath spot of sugarcane 4.
Dark purple midrib streaks associated with Phoma sorghina, Drechslera rostrata and D. australiensis were described from CoC. 776 collected from Nellore district. Phoma sorghina was reported as a new record on sugarcane from India 14. Drechslera (Helminthosporium) rostrata associated with brown stripe lesions on sugarcane was reported from Florida 15. D. australiensis isolated in the present work appears to be a new record on sugarcane.
Dark red linear lesions associated with Alternaria phragmospora, Curvularia lunata and Drechslera hawaiiensis were observed on the sugarcane variety Co. 6907, grown in Nellore district. D. hawaiiensis was reported in Argentina from sugarcane seedlings exhibiting blight in the nursery 16. Alternaria phragmospora isolated from dark red linear lesions described in the present work forms a new host record on sugarcane,
Dark red midribs associated with Alternaria alternata, Curvularia lunata var. aeria and Drechslera longirostrata were described on sugarcane variety Co. 62175, cultivated in Nellore district.
Alternaria(tenuis )alternata causing reddish discolouration of sugarcane midribs was reported from Taiwan 17.11 described severe blight of sugarcane associated with Alternaria tenuis and Drechslera (Helminthosporium) halodes. They stated that the symptoms produced by A. tenuis were quite distinct from the symptoms produced by D. halodes. However, both the fungi were found together in the blighted leaves, Curvularia lunata var. aeria isolated in the present work appears to be a new record on sugarcane.
Pathogenicity tests could, not be carried out with different fungi isolated from different types of leaf spots described in this dissertation programme, in order to identify the pathogen responsible for a particular type of leaf spot of sugarcane. However/ it may be considered that some of the fungi isolated and described here are potential, foliar parasites of sugarcane.
The following fungi viz., Curvularia tuberculata, C. pallescens, C. borreriae, C. robusta, C. lunata var. aeria, Drechslera holmii, D. australiensis and Alternaria phragmospora isolated from the minor leaf spots of different varieties of sugarcane, form new host records of this crop.